Human trials of Oxford coronavirus vaccine paused over ‘spinal-cord disease fears’
Leaked report says volunteer in UK was left unable to walk after receiving two doses
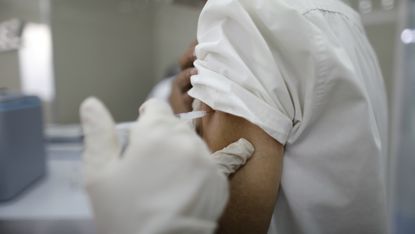
Human trials of the Oxford coronavirus vaccine are facing delays in the US after a participant suffered a rare neurological condition, according to a leaked document.
The AstraZeneca report, seen by CNN, shows that the decision to pause the trials in early September was made after a previously healthy 37-year-old woman who had received two doses developed transverse myelitis, an inflammation of the spinal cord that can cause paralysis.
The unidentified UK-based volunteer was given the jabs in June and August but was hospitalised after she started to suffer “difficulty walking, pain and weakness in her arms and a headache after tripping while running in September”, the Daily Mail reports.
Subscribe to The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
She is understood to have recovered fully.
The internal report by AstraZeneca, which owns the rights to the so-called AZD1222 vaccine, also reveals that the trials had been stopped before after another participant developed transverse myelitis.
“The first pause, in July, was not publicly revealed and the trial was restarted after it was determined the volunteer had multiple sclerosis, a condition that can cause the same neurological reaction,” says The Telegraph.
Following the latest hold-up, trials have resumed in the UK, Brazil, India and South Africa, but remain suspended in the US.
The “concerns around neurological side-effects” of vaccines are “especially sensitive in the US”, according to the newspaper, which notes that an emergency influenza vaccine issued in 1976 caused 450 cases, of which 30 were fatal, of Guillain-Barre syndrome, which also causes inflammation of the spinal cord.
The US Food and Drug Administration (FDA) and the National Institutes of Health (NIH) are reportedly “seeking to determine” what caused the case of transverse myelitis in the Covid trials before giving the green light for the testing to resume.
Create an account with the same email registered to your subscription to unlock access.
Sign up for Today's Best Articles in your inbox
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
-
 Georgia's new foreign influence bill
Georgia's new foreign influence billUnder the Radar Critics claim the 'Russian law' could stifle dissent and wreck the country's chances of joining the EU
By Elliott Goat, The Week UK Published
-
 'Making a police state out of the liberal university'
'Making a police state out of the liberal university'Instant Opinion Opinion, comment and editorials of the day
By Harold Maass, The Week US Published
-
 8 looming climate tipping points that imperil our planet
8 looming climate tipping points that imperil our planetThe Explainer New reports detail the thresholds we may be close to crossing
By Devika Rao, The Week US Published
-
 Covid four years on: have we got over the pandemic?
Covid four years on: have we got over the pandemic?Today's Big Question Brits suffering from both lockdown nostalgia and collective trauma that refuses to go away
By Chas Newkey-Burden, The Week UK Published
-
 The hollow classroom
The hollow classroomOpinion Remote school let kids down. It will take much more than extra tutoring for kids to recover.
By Mark Gimein Published
-
 Excess screen time is making children only see what is in front of them
Excess screen time is making children only see what is in front of themUnder the radar The future is looking blurry. And very nearsighted.
By Devika Rao, The Week US Published
-
 Covid-19: what to know about UK's new Juno and Pirola variants
Covid-19: what to know about UK's new Juno and Pirola variantsin depth Rapidly spreading new JN.1 strain is 'yet another reminder that the pandemic is far from over'
By Arion McNicoll, The Week UK Published
-
 Long-term respiratory illness is here to stay
Long-term respiratory illness is here to stayThe Explainer Covid is not the only disease with a long version
By Devika Rao, The Week US Published
-
 Covid inquiry: the most important questions for Boris Johnson
Covid inquiry: the most important questions for Boris JohnsonTalking Point Former PM has faced weeks of heavy criticism from former colleagues at the public hearing
By The Week Staff Published
-
 China's pneumonia cases: should we be worried?
China's pneumonia cases: should we be worried?The Explainer Experts warn against pushing 'pandemic panic button' following outbreak of respiratory illness
By Keumars Afifi-Sabet, The Week UK Published
-
 Vallance diaries: Boris Johnson 'bamboozled' by Covid science
Vallance diaries: Boris Johnson 'bamboozled' by Covid scienceSpeed Read Then PM struggled to get his head around key terms and stats, chief scientific advisor claims
By The Week UK Published