Coronavirus: how much protection can we expect from a future vaccine?
Oxford jab trials nearing completion - but experts dismiss hopes of ‘pandemic silver bullet’
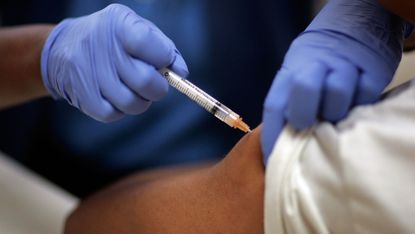
With countries worldwide heading back into lockdown, speculation is growing about when a potential vaccine may become available to help alleviate the need for harsh Covid-19 restrictions.
UK Vaccine Taskforce chair Kate Bingham told MPs this week that while the Oxford University vaccine may be ready within weeks, production delays mean the first batch may total no more than four million doses. And concerns over how long coronavirus antibodies last have raised questions about whether multiple jabs may be needed to maintain immunity.
So how much protection can we expect from the first Covid vaccines?
Subscribe to The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
When will a vaccine be ready?
The director of the Oxford University vaccine trial has said there is a “small chance” of the jab being ready before Christmas.
Professor Andrew Pollard told MPs on the Commons Science and Technology Committee on Wednesday that he was “optimistic” the trial could present late-stage results before the end of the year - a development that “by historical standards, is phenomenal”, says the BBC’s health and science correspondent James Gallagher.
“It would normally take years, if not decades, to develop a vaccine,” Gallagher adds. “Just to get to this point has been a remarkable and unprecedented feat, and data will arrive soon which will define our lives for years.”
However, most experts have suggested that early to mid 2021 is a more realistic rollout date.
Hundreds of potential vaccines are being tested in countries across the globe, with 154 candidates currently in pre-clinical trials, 21 in small-scale human safety trials, 12 in more expanded safety trials, and 11 in wider testing and effectivness assessments.
How well will it work?
Dr Anthony Fauci, the chief of the US National Institute of Health and Infectious Disease, has warned for months that a vaccine may not be a silver bullet. Addressing a webinar hosted by Brown University in August, he said: “We don't know yet what the efficacy might be. We don't know if it will be 50% or 60%.”
Other experts have also called for caution. In a paper published in The Lancet in September, Sri Lankan virologist Malik Peiris and Hong Kong epidemiologist Gabriel M. Leung write that “we cannot assume Covid-19 vaccines, even if shown to be effective in reducing severity of disease, will reduce virus transmission to a comparable degree.
“The notion that Covid-19-vaccine-induced population immunity will allow a return to pre-Covid-19 ‘normalcy’ might be based on illusory assumptions.”
The US Food and Drug Administration has said that “once a vaccine is shown to be safe and at least 50% effective, it could be approved for use in the US”, reports NPR.
But in order to get 40% of a population immune through vaccination with this level of efficacy, “you’re going to have to vaccinate 80% of the population”, Carlos del Rio, an infectious disease expert at Emory University, told the Washington D.C.-based news site. “So it’s not going to happen right away.”
Study results published by Imperial College London in October throw up a further problem. The research revealed that antibodies against the coronavirus declined rapidly in the UK population over the summer months.
The findings suggests that the protection provided by vaccines may not last, raising questions over whether multiple jabs may be needed to ensure immunity.
Read more: who would be first to get vaccinated in the UK?
Create an account with the same email registered to your subscription to unlock access.
Sign up for Today's Best Articles in your inbox
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
-
 Who actually needs life insurance?
Who actually needs life insurance?The Explainer If you have kids or are worried about passing on debt, the added security may be worth it
By Becca Stanek, The Week US Published
-
 Sexual wellness trends to know, from products and therapies to retreats and hotels
Sexual wellness trends to know, from products and therapies to retreats and hotelsThe Week Recommends Talking about pleasure and sexual health is becoming less taboo
By Theara Coleman, The Week US Published
-
 Is the AI bubble deflating?
Is the AI bubble deflating?Today's Big Question Growing skepticism and high costs prompt reconsideration
By Joel Mathis, The Week US Published
-
 Covid four years on: have we got over the pandemic?
Covid four years on: have we got over the pandemic?Today's Big Question Brits suffering from both lockdown nostalgia and collective trauma that refuses to go away
By Chas Newkey-Burden, The Week UK Published
-
 The hollow classroom
The hollow classroomOpinion Remote school let kids down. It will take much more than extra tutoring for kids to recover.
By Mark Gimein Published
-
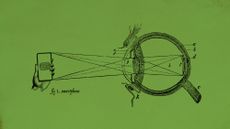 Excess screen time is making children only see what is in front of them
Excess screen time is making children only see what is in front of themUnder the radar The future is looking blurry. And very nearsighted.
By Devika Rao, The Week US Published
-
 Covid-19: what to know about UK's new Juno and Pirola variants
Covid-19: what to know about UK's new Juno and Pirola variantsin depth Rapidly spreading new JN.1 strain is 'yet another reminder that the pandemic is far from over'
By Arion McNicoll, The Week UK Published
-
 Long-term respiratory illness is here to stay
Long-term respiratory illness is here to stayThe Explainer Covid is not the only disease with a long version
By Devika Rao, The Week US Published
-
 Covid inquiry: the most important questions for Boris Johnson
Covid inquiry: the most important questions for Boris JohnsonTalking Point Former PM has faced weeks of heavy criticism from former colleagues at the public hearing
By The Week Staff Published
-
 China's pneumonia cases: should we be worried?
China's pneumonia cases: should we be worried?The Explainer Experts warn against pushing 'pandemic panic button' following outbreak of respiratory illness
By Keumars Afifi-Sabet, The Week UK Published
-
 Vallance diaries: Boris Johnson 'bamboozled' by Covid science
Vallance diaries: Boris Johnson 'bamboozled' by Covid scienceSpeed Read Then PM struggled to get his head around key terms and stats, chief scientific advisor claims
By The Week UK Published