Has Britain’s Covid response finally become ‘world beating’ - and did Brexit boost vaccine rollout?
Health secretary claims speedy approval of Pfizer jab is thanks to UK’s impending departure from EU
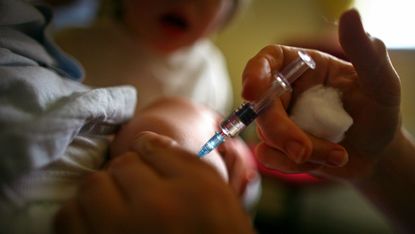
With the highest Covid death toll in Europe and the botched launch of the NHS Track and Trace app, Britain’s promised “world-beating” response to the pandemic has failed to live up to that billing since Boris Johnson made the pledge back in May.
“For once, the UK really was world-beating,” says Politico. But can the country maintain its lead - and what does Brexit have to do with it all?
How the vaccine was delivered
Subscribe to The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
The vaccine was authorised for emergency use by the UK’s Medicines and Healthcare products Regulatory Authority (MHRA) this week, under special powers granted to the executive agency.
The vaccine will be free but not compulsory, and will be delivered in three ways, the BBC reports. Patients will receive the jab at either a hospital, at GPs or pharmacists, or at vaccination centres that Health Secretary Matt Hancock says will be “a bit like” the Nightingales hospitals set up to add capacity to the NHS during the first wave of coronavirus.
About 50 hospitals are on stand-by, and vaccination centres in venues including conference centres or sports stadiums are being prepared.
The prime minister has hailed the vaccine approval as “unquestionably good news”, but also warned that the Covid immunisation rollout is “by no means the end of the story”.
Is jab victory thanks to Brexit?
Hancock and some other politicians have claimed that Brexit allowed the UK to approve the vaccine faster than any other country.
The health secretary told Times Radio yesterday that the UK was doing “all the same safety checks and the same processes, but we have been able to speed up how they’re done because of Brexit”.
Meanwhile, leader of the House of Commons and arch-Brexiteer Jacob Rees-Mogg, tweeted: “We could only approve this vaccine so quickly because we have left the EU.”
However, “both Downing Street and the UK’s medicines regulator have contradicted” that claim, says The Guardian. The goverment announcement of the vaccine approval “said it had taken place under a provision of the Human Medicines Regulations, passed in 2012, which permits the rapid licensing of medicines in the event of an emergency such as a pandemic”, the paper reports.
Education Secretary Gavin Williamson has also dismissed the notion that the UK won the vaccine race because of Brexit - but still struck a jingoistic note.
“I just reckon we’ve got the very best people in this country and we’ve obviously got the best medical regulators... much better than the French, much better than the Belgians have, much better than the Americans have,” Williamson told LBC radio.
“That doesn’t surprise me at all because we’re a much better country than every single one of them.”
Who will grant approval next?
The Telegraph reports that EU authorities have “set out a rough timeline for a decision” on the Pfizer/BioNTech vaccine, with the European Medicines Agency’s scientific committee for human medicines due to meet “to conclude its assessment by 29 December, at the latest”.
The US, meanwhile, is a little further behind. “Unlike many of its counterparts”, the US Food and Drug Administration has rules that “require the agency to complete an internal assessment and to consult an external advisory board prior to making a decision”, causing significant delays, says the newspaper.
China and Russia are developing and distributing their own vaccines, with Russia’s Sputnik V jab thought to be the most promising for either country so far.
Create an account with the same email registered to your subscription to unlock access.
Sign up for Today's Best Articles in your inbox
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
-
 Nigeria's worsening rate of maternal mortality
Nigeria's worsening rate of maternal mortalityUnder the radar Economic crisis is making hospitals unaffordable, with women increasingly not receiving the care they need
By Harriet Marsden, The Week UK Published
-
 'Elevating Earth Day into a national holiday is not radical — it's practical'
'Elevating Earth Day into a national holiday is not radical — it's practical'Instant Opinion Opinion, comment and editorials of the day
By Harold Maass, The Week US Published
-
 UAW scores historic win in South at VW plant
UAW scores historic win in South at VW plantSpeed Read Volkswagen workers in Tennessee have voted to join the United Auto Workers union
By Peter Weber, The Week US Published
-
 Covid four years on: have we got over the pandemic?
Covid four years on: have we got over the pandemic?Today's Big Question Brits suffering from both lockdown nostalgia and collective trauma that refuses to go away
By Chas Newkey-Burden, The Week UK Published
-
 The hollow classroom
The hollow classroomOpinion Remote school let kids down. It will take much more than extra tutoring for kids to recover.
By Mark Gimein Published
-
 Excess screen time is making children only see what is in front of them
Excess screen time is making children only see what is in front of themUnder the radar The future is looking blurry. And very nearsighted.
By Devika Rao, The Week US Published
-
 Car fatality rates are driving up
Car fatality rates are driving upThe Explainer The dang Covid pandemic made us worse drivers
By Devika Rao, The Week US Published
-
 Covid-19: what to know about UK's new Juno and Pirola variants
Covid-19: what to know about UK's new Juno and Pirola variantsin depth Rapidly spreading new JN.1 strain is 'yet another reminder that the pandemic is far from over'
By Arion McNicoll, The Week UK Published
-
 Long-term respiratory illness is here to stay
Long-term respiratory illness is here to stayThe Explainer Covid is not the only disease with a long version
By Devika Rao, The Week US Published
-
 Covid inquiry: the most important questions for Boris Johnson
Covid inquiry: the most important questions for Boris JohnsonTalking Point Former PM has faced weeks of heavy criticism from former colleagues at the public hearing
By The Week Staff Published
-
 China's pneumonia cases: should we be worried?
China's pneumonia cases: should we be worried?The Explainer Experts warn against pushing 'pandemic panic button' following outbreak of respiratory illness
By Keumars Afifi-Sabet, The Week UK Published