Scientists begin testing two coronavirus vaccines
Australian researchers reach ‘critical milestone’ in global Covid-19 battle
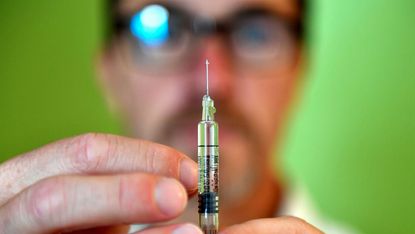
Scientists in Australia have begun testing two potential coronavirus vaccines, marking a “critical milestone” in the fight to bring the global pandemic under control.
The vaccines, made by Oxford University and US company Inovio Pharmaceutical, have been cleared for animal testing by the World Health Organization.
The first human trial for a coronavirus vaccine took place in the US last month, the BBC reports. But Australia’s Commonwealth Scientific and Industrial Research Organisation (CSIRO) says its tests will be the first pre-clinical trials of the vaccines using an animal model – in this case ferrets, which contract the coronavirus as humans do.
Subscribe to The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
The Guardian adds that approximately 35 companies and academic institutions around the world are currently racing to create a vaccine.
–––––––––––––––––––––––––––––––For a round-up of the most important stories from around the world - and a concise, refreshing and balanced take on the week’s news agenda - try The Week magazine. Start your trial subscription today –––––––––––––––––––––––––––––––
“Normally it takes about one-to-two years to get to this point and we’ve in fact shortened that to a period of a couple of months,” Rob Grenfell from CSIRO told reporters.
“This unprecedented speed is thanks in large part to early Chinese efforts to sequence the genetic material of Sars-CoV-2, the virus that causes Covid-19,” says The Guardian.
“China shared that sequence in early January, allowing research groups around the world to grow the live virus and study how it invades human cells and makes people sick.”
Another factor is access to existing research combating similar viruses, particularly severe acute respiratory syndrome (Sars) and Middle East respiratory syndrome (Mers).
According to Yahoo Finance, scientists elsewhere are also trialling existing drugs used to treat conditions such as Ebola, HIV, malaria and tuberculosis in order to establish if they could be used against the coronavirus.
“The ideal vaccine would be safe, easy to administer, simple and cheap to manufacture, and provide long-term protection against Covid-19,” write Kylie Quinn and Damian Purcell on The Conversation.
“But, to begin with, we’d even be happy with a vaccine that could reduce the amount of virus generated during a typical infection. If an infected person is making less virus, they are less likely to infect others. Less virus could also reduce the amount of damage caused by an infection in the patient.”
The first results from animal tests could be revealed as early as June, scientists say, and if successful, the vaccines could then be moved into clinical trials that could be conducted at labs elsewhere.
“At that point, the race to get the medicine into the general market could be accelerated, but experts warn it would still involve a minimum 18-month timeline to meet regulatory tests and standards,” says the BBC.
Create an account with the same email registered to your subscription to unlock access.
Sign up for Today's Best Articles in your inbox
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
-
 Post Office's Capture software to be reviewed over 'glitches'
Post Office's Capture software to be reviewed over 'glitches'Speed Read Solicitor representing accused postmasters says flaws in the IT system follow 'very similar pattern' to Horizon
By Arion McNicoll, The Week UK Published
-
 How would we know if World War Three had started?
How would we know if World War Three had started?Today's Big Question With conflicts in Ukraine, Middle East, Africa and Asia-Pacific, the 'spark' that could ignite all-out war 'already exists'
By Harriet Marsden, The Week UK Published
-
 Death Cafe: where people talk mortality over tea and cake
Death Cafe: where people talk mortality over tea and cakeWhy everyone's talking about The meet-ups are intended to offer a judgement-free and respectful space to discuss the end of life
By Chas Newkey-Burden, The Week UK Published
-
 Covid four years on: have we got over the pandemic?
Covid four years on: have we got over the pandemic?Today's Big Question Brits suffering from both lockdown nostalgia and collective trauma that refuses to go away
By Chas Newkey-Burden, The Week UK Published
-
 The hollow classroom
The hollow classroomOpinion Remote school let kids down. It will take much more than extra tutoring for kids to recover.
By Mark Gimein Published
-
 'Beyond belief': fears of asbestos return
'Beyond belief': fears of asbestos returnUnder the radar Attention is returning to the dangers of the carcinogenic substance
By Chas Newkey-Burden, The Week UK Published
-
 Excess screen time is making children only see what is in front of them
Excess screen time is making children only see what is in front of themUnder the radar The future is looking blurry. And very nearsighted.
By Devika Rao, The Week US Published
-
 Covid-19: what to know about UK's new Juno and Pirola variants
Covid-19: what to know about UK's new Juno and Pirola variantsin depth Rapidly spreading new JN.1 strain is 'yet another reminder that the pandemic is far from over'
By Arion McNicoll, The Week UK Published
-
 Long-term respiratory illness is here to stay
Long-term respiratory illness is here to stayThe Explainer Covid is not the only disease with a long version
By Devika Rao, The Week US Published
-
 Covid inquiry: the most important questions for Boris Johnson
Covid inquiry: the most important questions for Boris JohnsonTalking Point Former PM has faced weeks of heavy criticism from former colleagues at the public hearing
By The Week Staff Published
-
 China's pneumonia cases: should we be worried?
China's pneumonia cases: should we be worried?The Explainer Experts warn against pushing 'pandemic panic button' following outbreak of respiratory illness
By Keumars Afifi-Sabet, The Week UK Published