Ebola: experimental vaccine trial an 'unqualified success'
Early human tests suggest the drug is safe and may help immune systems fight Ebola, but more trials are needed
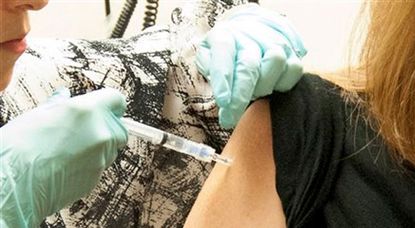
The first human trial of an experimental Ebola vaccine has delivered promising results, according to the US National Institute of Health (NIH).
"On safety and on the ability to produce an appropriate immune response we can call this trial an unqualified success," Dr Anthony Fauci of the NIH told the BBC.
However, he stressed that this was only the early part of the first phase of the trial and much more testing was required to establish the vaccine's long-term safety and efficacy.
Subscribe to The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
There is currently no vaccine or cure for Ebola and the unprecedented scale of the current outbreak in West Africa has prompted pharmaceutical companies and health organisations to fast track the development of safe and effective treatments.
The vaccine has been jointly produced by the NIH and the British pharmaceutical company GlaxoSmithKline (GSK) and is one of three currently in development.
In the first phase of the trial, all 20 American volunteers who received the vaccine developed antibodies to the virus and none suffered any major side effects. However, some patients did develop a fever in response to the drug, but it "resolved within one day", according to scientists.
GSK has reportedly made a request for an indemnity agreement to protect itself against unforeseen side effects that could develop in the future.
"It's important to remember that these data are the first piece in the jigsaw and we're continuing to gather other important information," Dr Moncef Slaoui, chairman of global vaccines at GSK told The Guardian.
If the results of the following phases of the trial are positive, thousands of volunteers, including healthcare workers, will receive the vaccine in West Africa in early 2015, he said.
Almost 5,700 people are known to have died from the disease and almost 15,000 have been infected across the region, though actual figures are believed to be much higher.
Create an account with the same email registered to your subscription to unlock access.
Sign up for Today's Best Articles in your inbox
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
-
 'A direct, protracted war with Israel is not something Iran is equipped to fight'
'A direct, protracted war with Israel is not something Iran is equipped to fight'Instant Opinion Opinion, comment and editorials of the day
By Harold Maass, The Week US Published
-
 Today's political cartoons - April 17, 2024
Today's political cartoons - April 17, 2024Cartoons Wednesday's cartoons - political anxiety, jury sorting hat, and more
By The Week US Published
-
 Arid Gulf states hit with year's worth of rain
Arid Gulf states hit with year's worth of rainSpeed Read The historic flooding in Dubai is tied to climate change
By Peter Weber, The Week US Published
-
 Dengue hits the Americas hard and early
Dengue hits the Americas hard and earlySpeed Read Puerto Rico has declared an epidemic as dengue cases surge
By Peter Weber, The Week US Published
-
 How happy is Finland really?
How happy is Finland really?Today's Big Question Nordic nation tops global happiness survey for seventh year in a row with 'focus on contentment over joy'
By Harriet Marsden, The Week UK Published
-
 How Tehran became the world's nose job capital
How Tehran became the world's nose job capitalUnder the radar Iranian doctors raise alarm over low costs, weak regulation and online influence of 'Western beauty standards'
By Harriet Marsden, The Week UK Published
-
 Africa's renewed battle against female genital mutilation
Africa's renewed battle against female genital mutilationUnder the radar Campaigners call for ban in Sierra Leone after deaths of three girls as coast-to-coast convoy prepares to depart
By Harriet Marsden, The Week UK Published
-
 Argentina: the therapy capital of the world
Argentina: the therapy capital of the worldUnder the radar Buenos Aires natives go hungry to pay for psychoanalysis, amid growing instability, anxiety – and societal acceptance
By Harriet Marsden, The Week UK Published
-
 Does declining birth rate spell doom for Britain?
Does declining birth rate spell doom for Britain?Today's Big Question Ageing population puts pressure on welfare state, economy and fabric of society, while fertility is rising on populist agendas
By Harriet Marsden, The Week UK Published
-
 How a new blood test could revolutionise sepsis diagnosis
How a new blood test could revolutionise sepsis diagnosisThe Explainer Early results from ongoing trial suggest faster identification of deadly condition is possible
By The Week Staff Published
-
 2023: the year of the loneliness epidemic
2023: the year of the loneliness epidemicthe explainer Loneliness isn't a new problem. But fighting it became a global health priority in 2023.
By Theara Coleman, The Week US Published